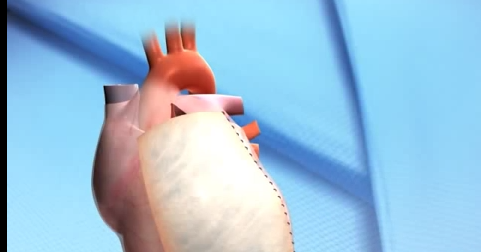
CorMatrix Cardiovascular secures FDA clearance for its Tyke biomaterial.
CorMatrix Cardiovascular has announced FDA 510(k) for its Tyke extracellular matrix biomaterial for use in neonates and infants.

CorMatrix Cardiovascular has announced FDA 510(k) for its Tyke extracellular matrix biomaterial for use in neonates and infants.